DISCOVER
CT-SCOUT™

Success is when the time to access an innovative treatment is reduced.
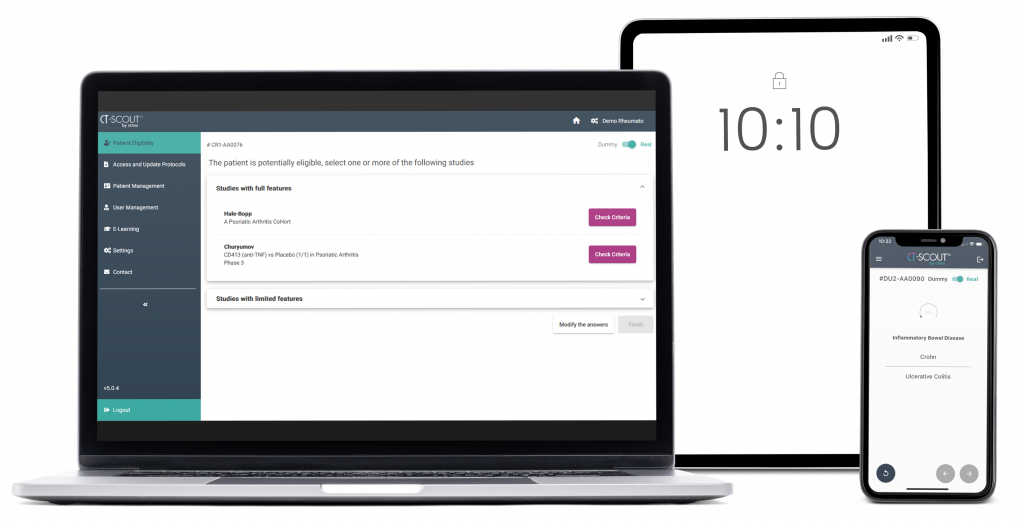
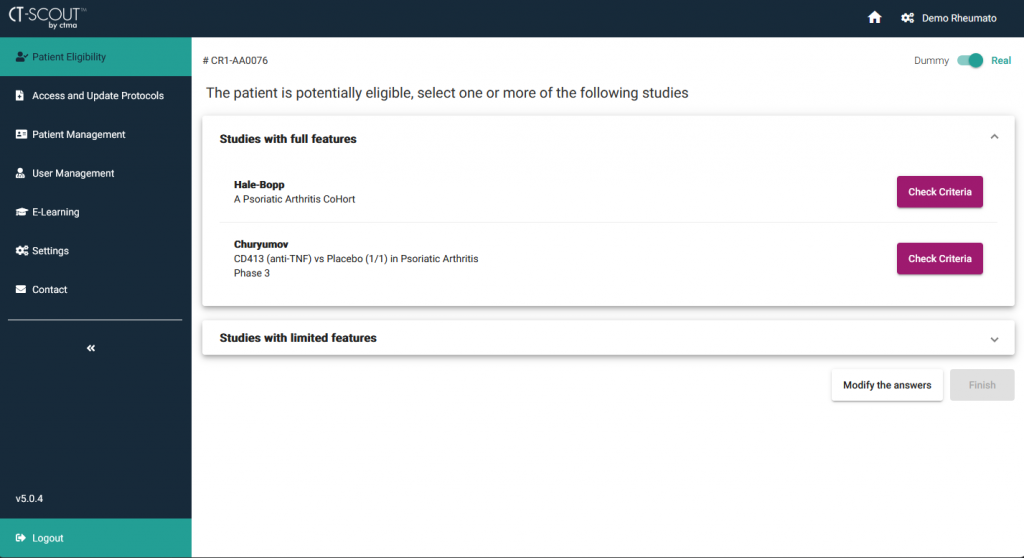
CT-SCOUT™ is a multi-device application aiming at facilitating the recruitment of patients in clinical studies...
It assists the investigational team in detecting the studies for which a patient is potentially eligible according to his/her current health conditions.
The digital solution to boost patient recruitment in clinical trials
Transforms all physician into real time recruiters
Anywhere, anytime
On any device
Easy to use
CT-SCOUT™ unique features
1. An efficient algorithmic model
2. Real-time detection
3. Simplified
process
Therapeutic Expertise
Gastroenterology
Crohn disease
Ulcerative colitis
Dermatology
Psoriasis
Atopic dermatitis
Hidradenitis suppurativa
Rheumatology
Rheumatoid arthritis
Ankylosing spondylitis
Psoriatic arthritis
200 +
studies registered (industrial & academic)
1000 +
users registered
170 +
centers in various countries (Belgium, Canada, France, Germany, Israel, Italy, Netherlands, Spain, Switzerland, UK…)
7000 +
Patients detected
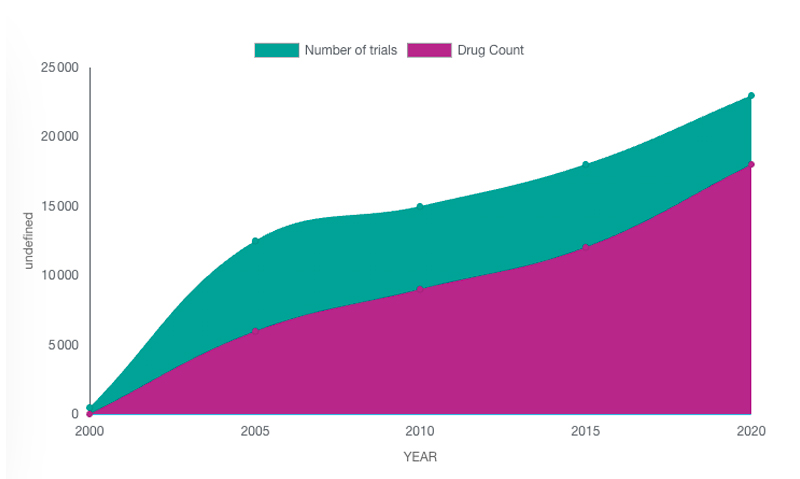
Figure: Total R&D/clinical trials pipeline size, by year 2000-2022
They are so many drugs in development that it is extremely challenging to finds the way to one of them, as a patient or as a site, or to secure a high visibility on one of them for a Sponsor. CT-SCOUT™and ToTem4me facilitate the match between the patients and the studies.

50%
OF STUDIES FAIL TO MEET PATIENT RECRUITMENT DEADLINES
However, millions of patients visit clinical sites every day.
CT-SCOUT™ help investigators identify the ones that could benefit from a clinical study.

80%
OF THE PATIENTS WOULD WELCOME THE OPPORTUNITY TO PARTICIPATE TO A CLINICAL STUDY
Patients have become actors of their treatment conditions.
ToTem4me help them identify potential opportunities through clinical studies.
0.02%
OF THE PATIENTS ARE BEING OFFERED TO PARTICIPATE TO A CLINICAL STUDY
But 80% of patients would accept to participate t a clinical study.
CT-SCOUT™ contributes to increase the percentage of patients considered for clinical research.

2
WEEKS SET-UP TIME
No more than 2 weeks are required to get CT-SCOUT™
or ToTem4me deployed for a clinical study
As a summary
Assist investigational teams in their clinical research activities.
Shorten clinical trials by boosting detection of patients potentially eligible to clinical trials.
Allow patients’ earlier access to innovative and life-saving treatment.
LATEST NEWSOUR NEWS >
April 30, 2026
Applied Clinical Trials Online Payment experience—including how, when, and with what level of transparency sites are compensated—is rapidly emerging as a defining factor in the success of clinical trials. Once dismissed as a simple operational nuisance, payments are now recognized as a critical component for shaping site relationships and study outcomes. For years, sites had […]
April 30, 2026
Med City News Certain features, commonplace on most e-commerce platforms, can help transform clinical trial search databases. Digital databases for clinical trial searches have already changed healthcare accessibility, bringing critical information about clinical trials to the patients who need it – and to the physicians who care for them. But these databases are also enormous […]
April 30, 2026
Med City News LLMs bring great potential to help the healthcare industry center care around patients’ needs by improving communication, access, and engagement. However, LLMs also present significant challenges associated with privacy and bias that also must be considered. Large language models (LLMs) have generated buzz in the medical industry for their ability to pass […]



